
The information provided on this Site is for communication, information exchanges and publications. Your access to and use of this Site is subject to the following terms and conditions, and all applicable laws and regulations. If you do not meet or comply with those, you may not use the Site.
Terms of Use
When browsing and using this Site, you will solely take all the consequences and risks, as with take all necessary precautions. This Site will not be responsible for any risks and consequences incurred by you.
In the process of accessing to and using this Site, you will not delete, modify or add any data or applications stored, disposed or transmitted in this Site; Furthermore, you will not use this Site to endanger the national website, disclose national secrets, infringe upon any national, social, collective or personal legal rights.
You will browse and use this Site legally, and agree not to use this Site in an Illegal, malicious, threatening, fraudulent, defamatory, or racially discriminatory way, nor widespread any information unfavourable to Porton Pharma Solutions Ltd. (herein after referred to as “Porton”). Or otherwise, Porton will retain the right to sue and claim for compensation.
Limitation of Liabilities
This Site will endeavour to provide the latest information, but will not expressively or impliedly guarantee the accuracy, timeliness, reliability, completeness, marketability and fitness for specific purposes. And this site will not be responsible for any liabilities arising out of.
This Site will not ensure that any functions do not suspend, omit or mistake, and will not guarantee that such deficiencies will be remedied promptly. Moreover, this Site will not guarantee the services and related servers provided does not involve any computer virus or harmful plug-ins. And this site will not be responsible for any liabilities with respect to such losses.
This Site and the information contained therein shall not constitute an offer or offer for the sale of any securities, or any official and formal disclosure form of company information. You, may search for the company information through China Securities Journal, Securities Times or other relative websites of stock changes. Any materials, information or viewpoints will not be deemed as the comments or suggestions of purchasing or selling stocks. You will bear all the liabilities and consequences if you purchase or sell securities relying on any materials or information.
This Site specially disclaims any liabilities in connection with the damages or losses when using or browsing, no matter any direct, indirect, incidental, punitive, secondary or other damages etc. caused by any factors, including but not limited to any failure, error, omission, interruption, deletion, defect, delay or alteration, any computer, program or document virus, network failure or force majeure etc.
Copyright
This Site and all designs, appearances, pictures, words, expressions and all those combinations under this Site belong to Porton, which are under the protection and regulation of the Copyright Law of People’s Republic of China, and relevant laws and regulations as well as international treaties.
This Site reserves the right to change or modify the content at any time, including but not limited to the appearance, style, format, content, as well as the products and services provided herein. You hereby agree to accept all modifications without any objection.
Without Prior consent of this Site, you are not allowed to copy, modify, distribute, disseminate, display, reproduce, publish, illegally use, reprint or do reverse engineer to any content of this site, or link this site to another site.
Trademark
Porton owns all trademarks of the products or services covered by (including but not limited to trade name, product decoration, services or product logos etc.). You may not copy, imitate or use them in whole or in part, and may not display or promote the trademarks under this Site in any circumstances
Privacy
You voluntarily leave your personal information for future cooperation with Porton, and agree Porton to use your personal information in compliance with laws and regulations. And Porton will respect your personal privacy and deal with your personal information, by abiding by laws and regulation of People’s Republic of China on network users’ personal information protection.
Law Application
This Legal Announcement and your use of this Site will be subject to the laws and regulations of China.
Severability
Any deficiencies or invalidity in any terms hereof shall not affect the validity of other terms hereof.
Explanation
This Site enjoys the right of final explanation regarding this Announcement.
©2023 Porton Advanced Solution Ltd. All Rights Reserved.
Porton Advanced Solutions is a subsidiary of Porton Pharma Solutions Ltd.
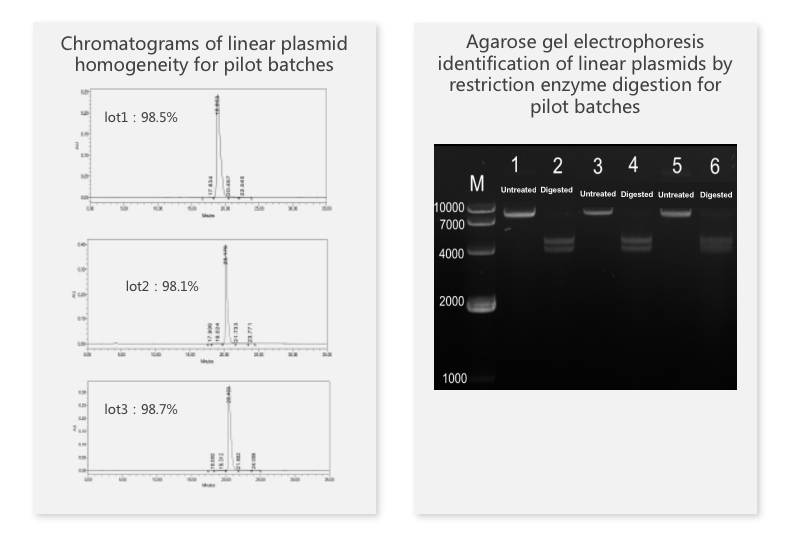



The repeatability study of selected samples at certain dilution levels showed great repeatability with a CV value of less than 10%.
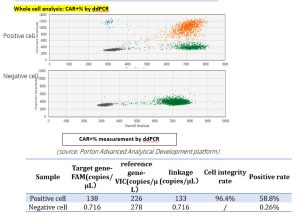
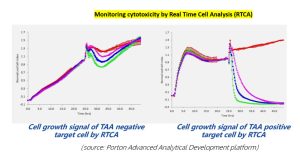
Case study

Assays

Case Study
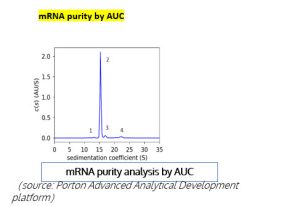
mRNA purity –AUC effectively distinguishes the different components in an mRNA sample, such as truncated mRNA (Peak 1), monomeric mRNA (Peak 2), long-chain mRNA, and mRNA aggregates (Peak 3& 4).
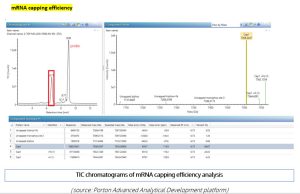
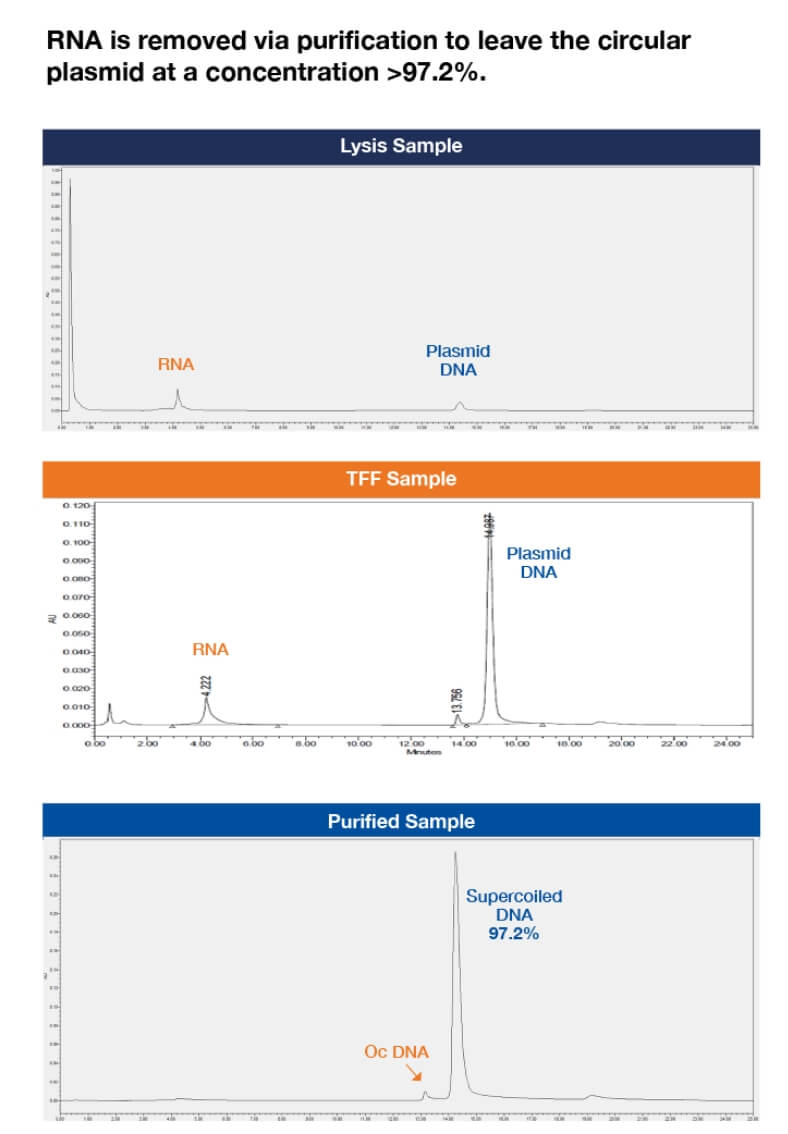
Figure 8
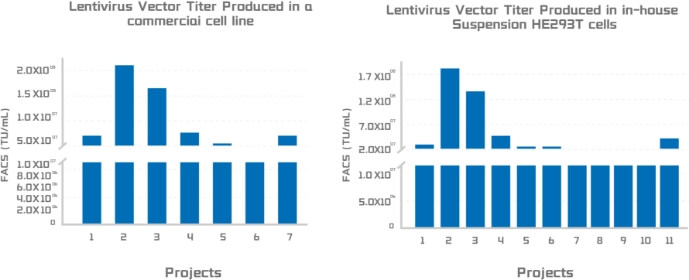
Lentivirus titer produced in two suspension cell production system:
A. Viral titer from a leading commercial packaging system. B. Viral titer from Porton Advanced’s PTLV-SMART packaging system.
Figure 4
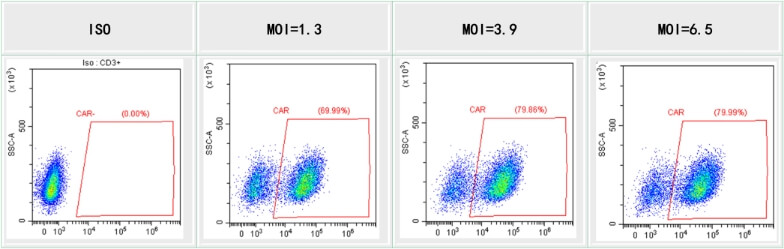
T cell transduction with lentivirus produced using our HEK293T system. T cells are infected with lentivirus at different MOI. CAR-T cell percentages were measured by FACS.
Figure 20
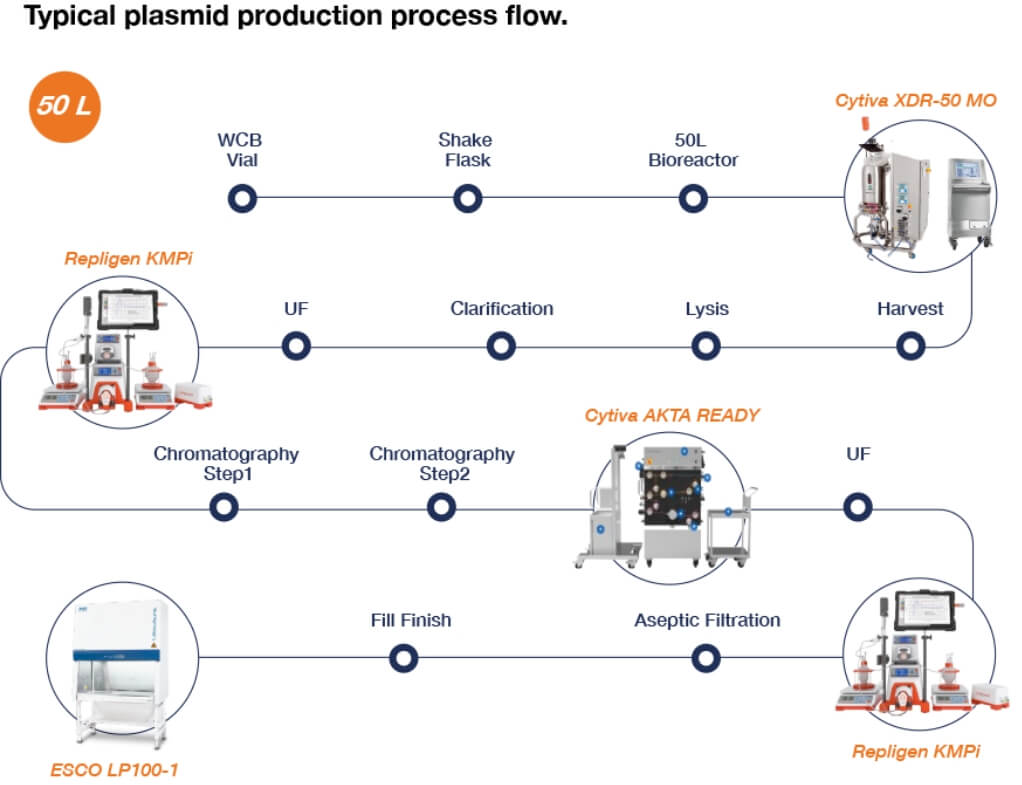
Figure 7

Figure 5
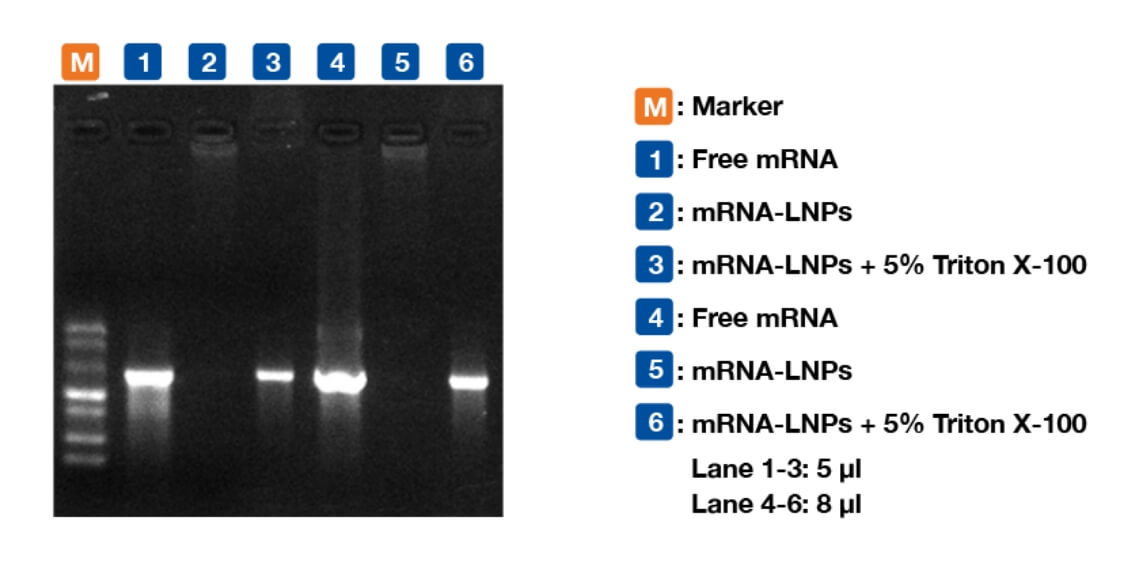
Figure 6

An AAV variant from our library screening showed that the virus was able to package a 6.1kb genome.
Figure 19

AAV variants enriched in different tissues during library screening. AAV libraries were prepared and injected into the hosts. After two weeks, tissues were harvested and viral genomes were isolated for PacBio sequencing. Viral genomes were clustered against different tissues.
Figure 18
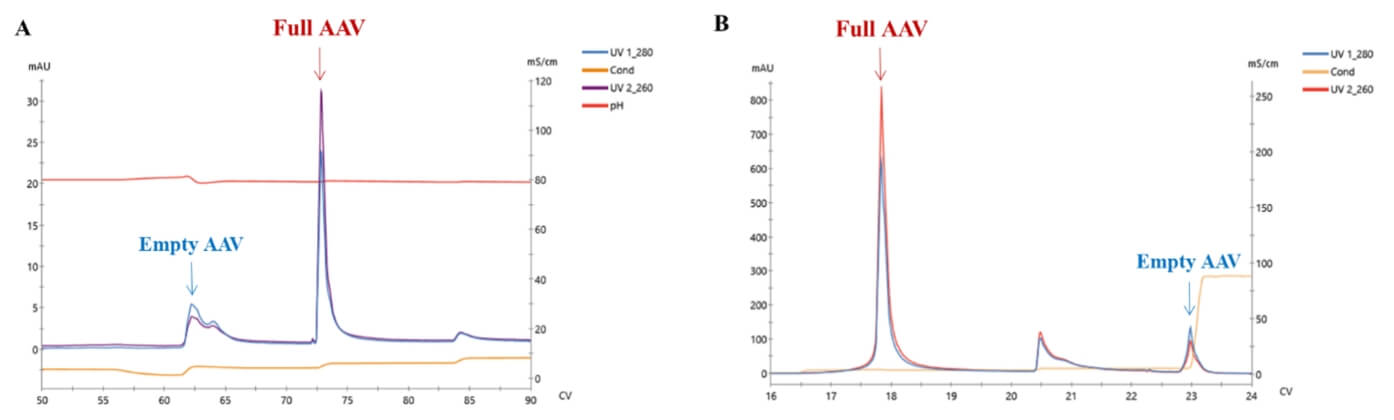
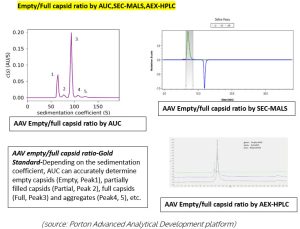
Separation of empty and full rAAV by anion chromatography. A: AEX result of rAAV5; B: AEX result of rAAV9
Figure 3
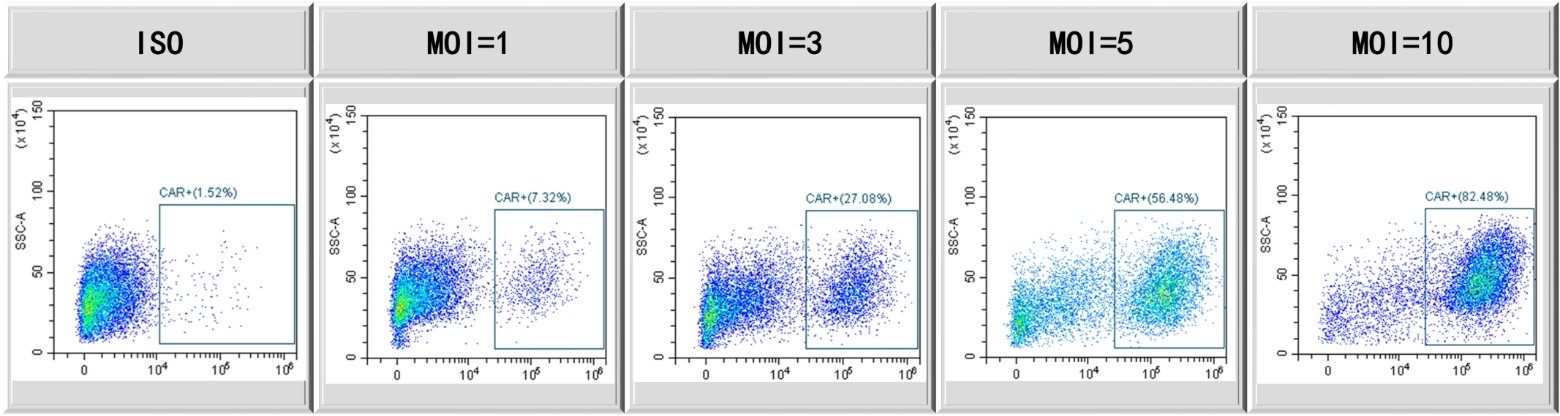
CAR expression with escalated MOI using PTLV-SMART platform. Activated T cells transduced with escalated MOI, and FACS employed to demonstrate CAR expression post-7 days culture in G-Rex.
Figure 1
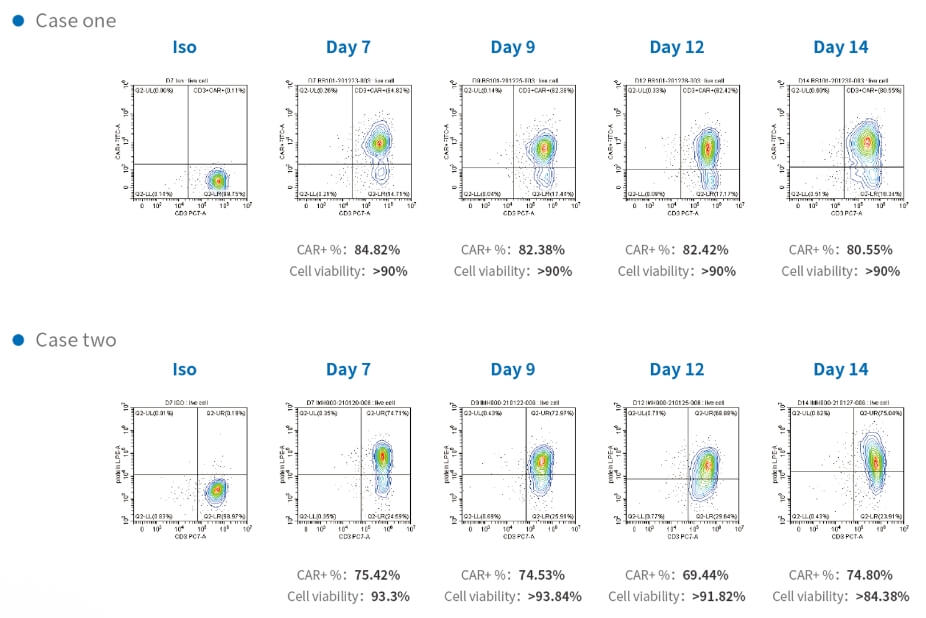
Time course of CAR transduction rates of CD3 T cells and cell viability during manufacturing in two cases. T-cells transduced with lentivirus analyzed by flow cytometry to detect CAR-positive or Protein L-positive cell ratio. T-cells expressed CAR stably in the period of CAR-T cell growth with high cell viability.
Figure 2