The analytical development and quality control services provided by Porton Advanced are established via seven different functional platforms: physicochemical, characterization, microbiology, molecular studies, biochemical, cell function, and FACS. These functional platforms support the precision testing of Advanced Therapy Medicinal Products(ATMPs), specializing in various analytical categories, such as general, purity, content, identity, integrity, potency, and safety testing. They provide differentiated analytical offerings that ensure accurate evaluation and characterization of advanced therapies.
Each platform’s expertise aids in safeguarding the safety, efficacy, and overall success of CGT products.
Physicochemical and Characterization assays play a crucial role in the development and manufacturing of CGT. These assays encompass a wide range of techniques and testing that assess the physical and chemical attributes of therapeutic products. This includes determining the identity, purity, potency, stability, and structural integrity of CGT products.
Our physicochemical analysis involves techniques such as liquid chromatography, mass spectrometry, spectroscopy, capillary electrophoresis, AUC, etc. These techniques enable the identification and quantification of critical components, impurities, and degradation in CGT products.

CASE STUDY
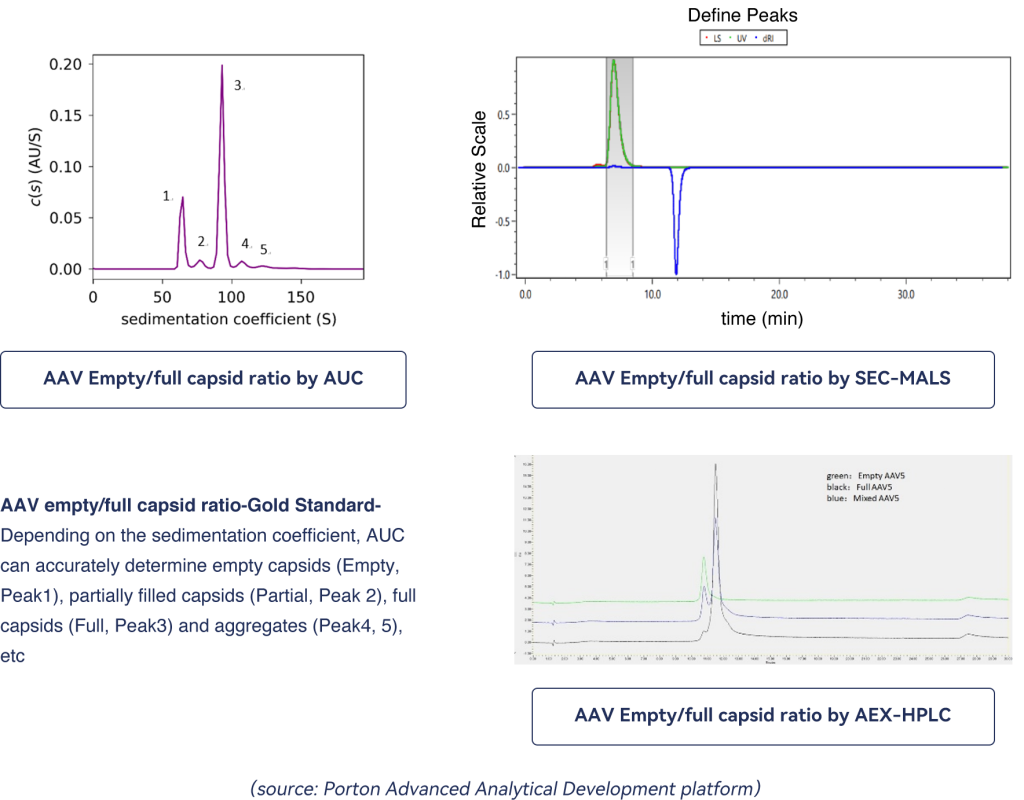
mRNA purity –AUC effectively distinguishes the different components in an mRNA sample, such as truncated mRNA (Peak 1), monomeric mRNA (Peak 2), long-chain mRNA, and mRNA aggregates (Peak 3& 4).

CASE STUDY
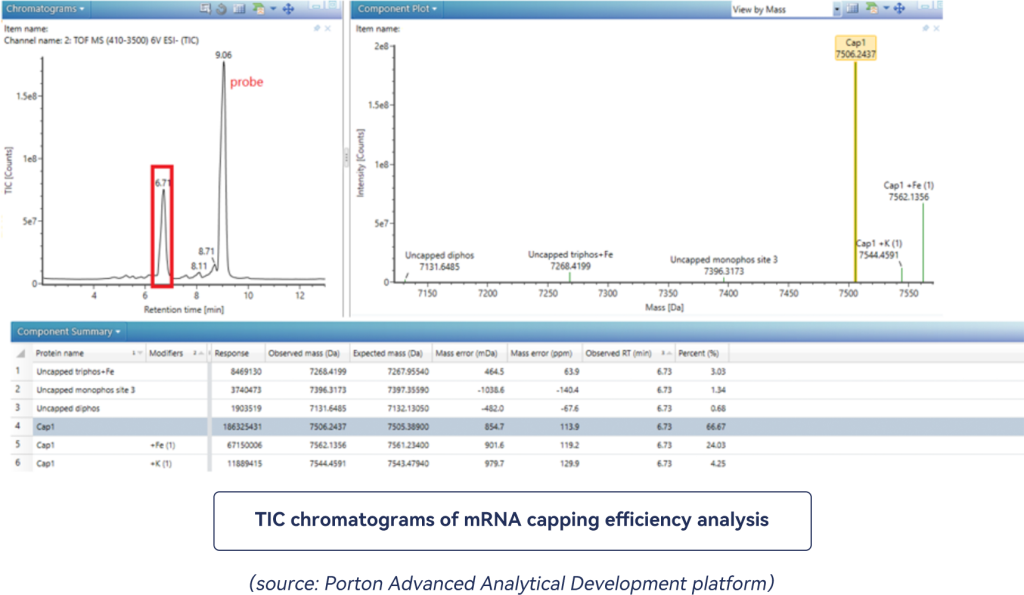
The Porton Advanced Characterization Platform utilizes LC-MS to analyze the capping ratio of the mRNA samples for the co-transcriptional capping reaction. Example:
The Rt 6.71 min in the left figure corresponds to the oligonucleotide fragments obtained by enzyme digestion. The 5′ capping ratio is obtained by quantifying the peak area of the capped or uncapped protein components.
Our microbiology team looks after cell bank testing, general microbiology testing, safety testing, such as sterility and endotoxin. For the sterility test, we implement the traditional method according to USP 71, and the rapid method for microbial detection using BD BACTEC FX40. Our team has completed alternative method validation, which decreases the turnaround time significantly.
For our molecular studies, we are equipped with ddPCR, gel doc gel imaging system, qPCRs, and PCRs of different models. These enable us to explore the relevant technologies including AGE, western blotting, qPCR, and ddPCR to establish methods such as nucleic acid identification, protein identification, residual DNA, copy number, and safety tests. Our safety tests include mycoplasma, sterility tests, RCL, and detection on endogenous/adventitious viruses.
CASE STUDY

source:Porton Advanced Analytical Development platform
As seen in the results above, by using ddPCR, the Porton Advanced molecular platform was able to detect the genome titers of rAAV samples at various dilutions.
CASE STUDY
The repeatability study of selected samples at certain dilution levels showed great repeatability with a CV value of less than 10%.
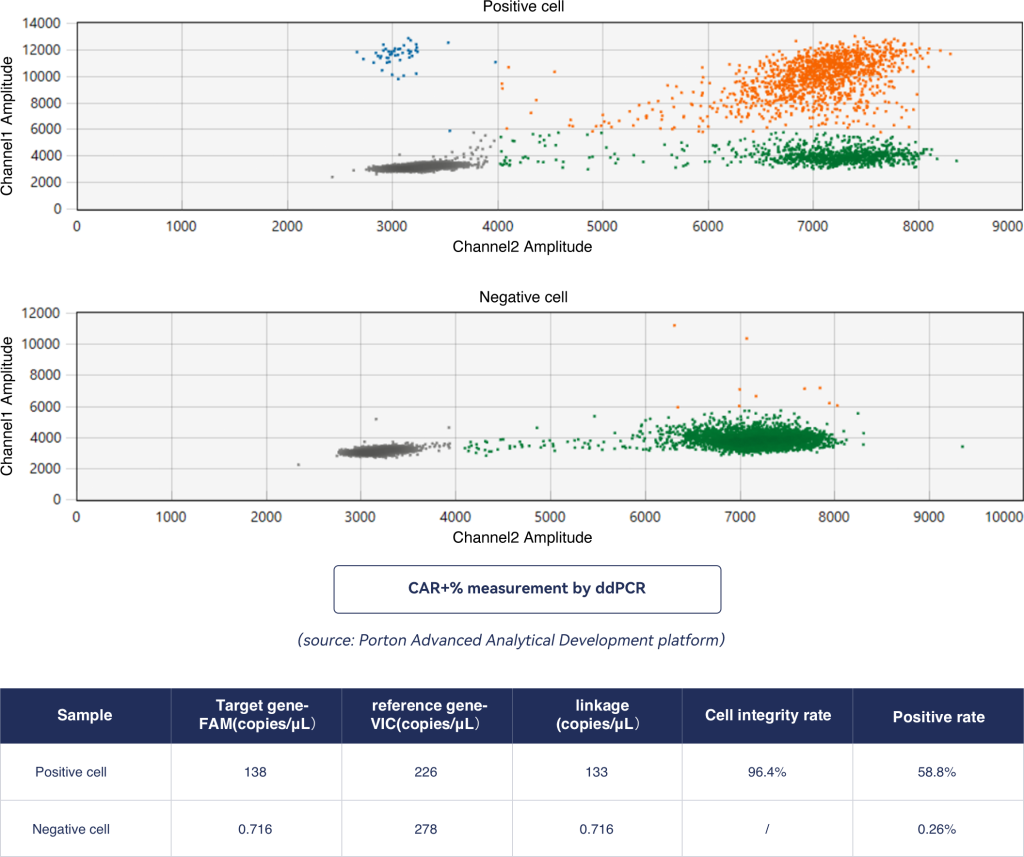
Analysis of Car+ cells using ddPCR presented strong cell integrity and stable CAR+ results.
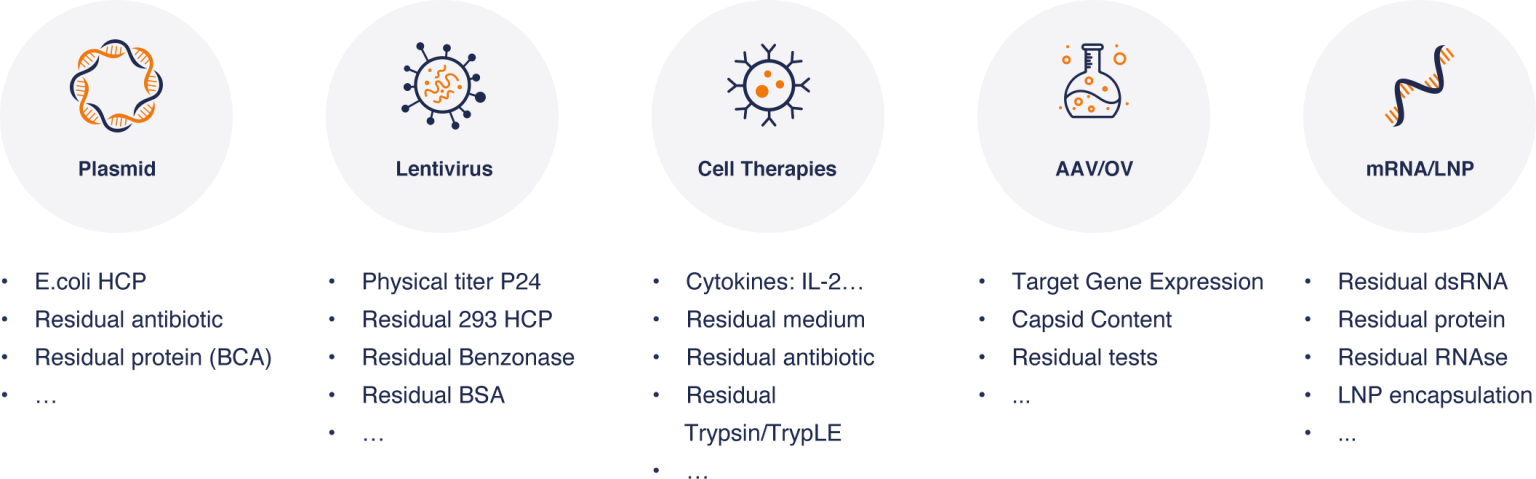
The biochemical testing platform covers an array of sample matrixes for plasmids, lentivirus, Car-Ts, and mRNAs (including LNP). Our platform offers a wide range of residual & impurity tests such as residual medium, residual antibiotic, residual medium, etc.
Summary of Reference Test Data
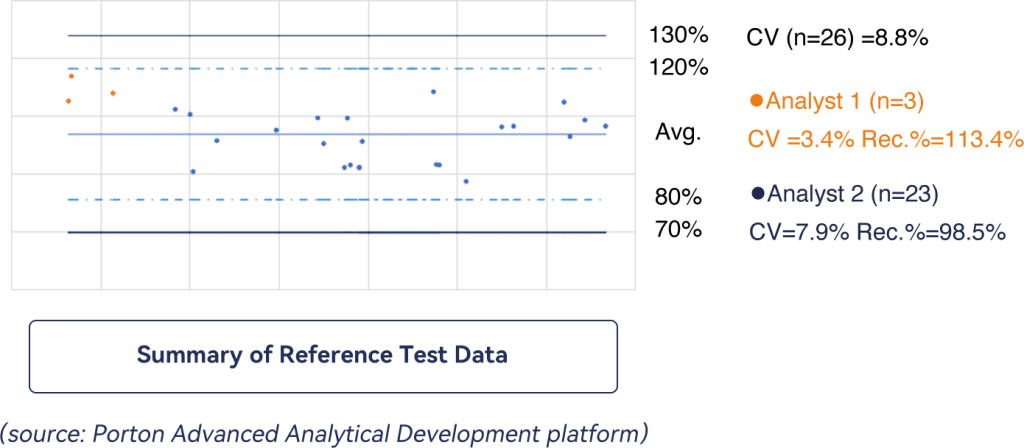
The team collects in-house reference data regularly. Data sets from different analysts proved accurate with a recovery rate of 80-120% and a repeatability precision with CV well below 10%.
Porton Advanced has a specific team who oversees cell functional tests. We spend a substantial amount of time and effort in exploring the cytotoxicity analysis, as well as other potency tests including IFN-y secretion assay. For example, when using RTCA (real-time cell analysis), the cell growth transforms into an electrical signal that monitors the target cell cytolysis in real time with higher sensitivity and precision. Alternatively, the FACS method labels the target cell with CFSE, enabling the measurement of non-adherent cells to guarantee results in 2 days. There is a lower cytotoxicity of the staining reagent and a lower baseline.
CASE STUDY

FACS (Fluorescence-Activated Cell Sorting) solutions play a crucial role in aiding the identification, isolation, and sorting of specific cell populations based on their unique fluorescent properties. Our robust in-house FACS based assays allow for a wider application, from the research and quality control stages during manufacturing and commercialization, all the way to ensuring product consistency and purity. This is done by verifying the identity and viability of cells, detecting contaminating cells, monitoring transduction efficiency, and confirming vector integration. Our team is skilled at precise cell population isolation, accurate phenotypic characterization, and stringent quality control, ensuring the production of safe and effective CGT that holds immense potential.
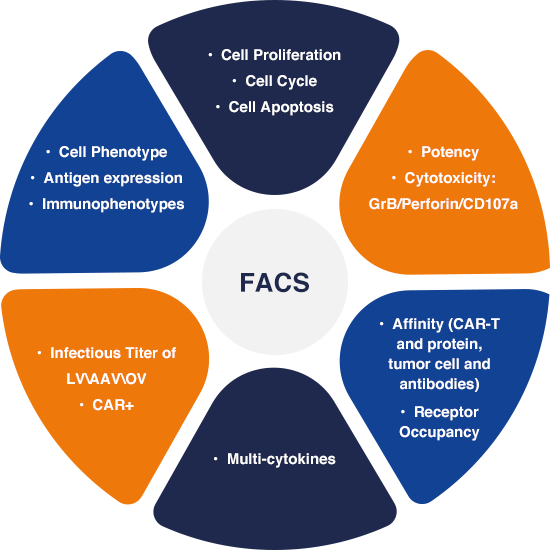
CASE STUDY
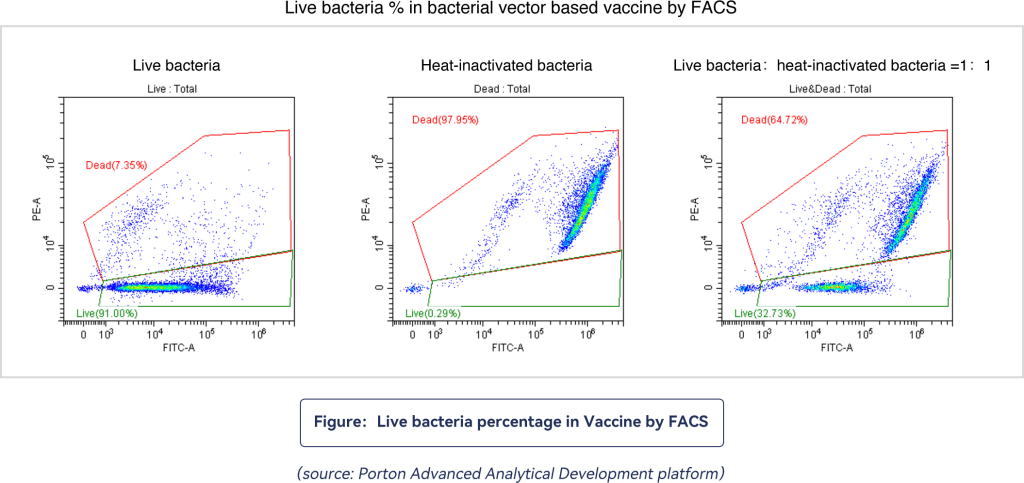
The FACS platform uses flow cytometry to detect the percentage of live bacteria in the intermediate process or preparation of Listeria.
Figure 1 shows that when a small number of dead bacteria is combined with PI in a live bacterial sample, red fluorescence is displayed. Green Fluorescence is mostly emitted when the bacteria is combined with SYTO9 dye. However, when the heat-inactivated bacterial samples are combined with PI due to cell membrane damage, a Red fluorescence is produced. In the 1:1 mixed sample of live bacteria and heat-inactivated bacteria, the detected red and green fluorescent signals are more visible.
FACS (Fluorescence-Activated Cell Sorting) solutions play a crucial role in aiding the identification, isolation, and sorting of specific cell populations based on their unique fluorescent properties. Our robust in-house FACS based assays allow for a wider application, from the research and quality control stages during manufacturing and commercialization, all the way to ensuring product consistency and purity. This is done by verifying the identity and viability of cells, detecting contaminating cells, monitoring transduction efficiency, and confirming vector integration. Our team is skilled at precise cell population isolation, accurate phenotypic characterization, and stringent quality control, ensuring the production of safe and effective CGT that holds immense potential.
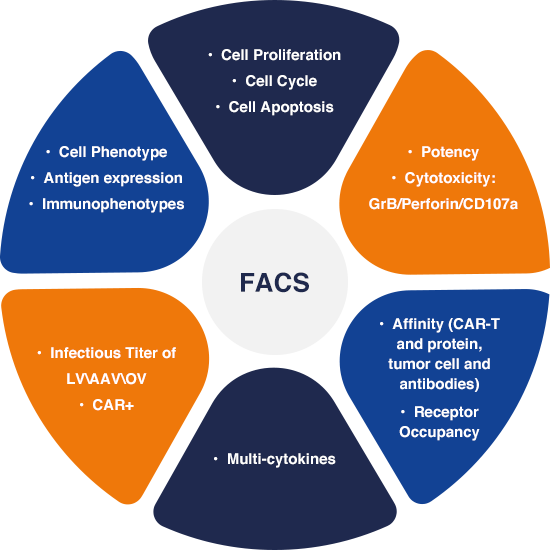
Physicochemical and Characterization assays play a crucial role in the development and manufacturing of CGT. These assays encompass a wide range of techniques and testing that assess the physical and chemical attributes of therapeutic products. This includes determining the identity, purity, potency, stability, and structural integrity of CGT products.
Our physicochemical analysis involves techniques such as liquid chromatography, mass spectrometry, spectroscopy, capillary electrophoresis, AUC, etc. These techniques enable the identification and quantification of critical components, impurities, and degradation in CGT products.

CASE STUDY

CASE STUDY
CASE STUDY
CASE STUDY
Physicochemical and Characterization assays play a crucial role in the development and manufacturing of CGT. These assays encompass a wide range of techniques and testing that assess the physical and chemical attributes of therapeutic products. This includes determining the identity, purity, potency, stability, and structural integrity of CGT products.
Our physicochemical analysis involves techniques such as liquid chromatography, mass spectrometry, spectroscopy, capillary electrophoresis, AUC, etc. These techniques enable the identification and quantification of critical components, impurities, and degradation in CGT products.

CASE STUDY

CASE STUDY
CASE STUDY
CASE STUDY
Physicochemical and Characterization assays play a crucial role in the development and manufacturing of CGT. These assays encompass a wide range of techniques and testing that assess the physical and chemical attributes of therapeutic products. This includes determining the identity, purity, potency, stability, and structural integrity of CGT products.
Our physicochemical analysis involves techniques such as liquid chromatography, mass spectrometry, spectroscopy, capillary electrophoresis, AUC, etc. These techniques enable the identification and quantification of critical components, impurities, and degradation in CGT products.

CASE STUDY

mRNA purity –AUC effectively distinguishes the different components in an mRNA sample, such as truncated mRNA (Peak 1), monomeric mRNA (Peak 2), long-chain mRNA, and mRNA aggregates (Peak 3& 4).

CASE STUDY
The Porton Advanced Characterization Platform utilizes LC-MS to analyze the capping ratio of the mRNA samples for the co-transcriptional capping reaction. Example:
The Rt 6.71 min in the left figure corresponds to the oligonucleotide fragments obtained by enzyme digestion. The 5′ capping ratio is obtained by quantifying the peak area of the capped or uncapped protein components.

Physicochemical and Characterization assays play a crucial role in the development and manufacturing of CGT. These assays encompass a wide range of techniques and testing that assess the physical and chemical attributes of therapeutic products. This includes determining the identity, purity, potency, stability, and structural integrity of CGT products.
Our physicochemical analysis involves techniques such as liquid chromatography, mass spectrometry, spectroscopy, capillary electrophoresis, AUC, etc. These techniques enable the identification and quantification of critical components, impurities, and degradation in CGT products.

CASE STUDY

mRNA purity –AUC effectively distinguishes the different components in an mRNA sample, such as truncated mRNA (Peak 1), monomeric mRNA (Peak 2), long-chain mRNA, and mRNA aggregates (Peak 3& 4).

CASE STUDY
The Porton Advanced Characterization Platform utilizes LC-MS to analyze the capping ratio of the mRNA samples for the co-transcriptional capping reaction. Example:
The Rt 6.71 min in the left figure corresponds to the oligonucleotide fragments obtained by enzyme digestion. The 5′ capping ratio is obtained by quantifying the peak area of the capped or uncapped protein components.

Physicochemical and Characterization assays play a crucial role in the development and manufacturing of CGT. These assays encompass a wide range of techniques and testing that assess the physical and chemical attributes of therapeutic products. This includes determining the identity, purity, potency, stability, and structural integrity of CGT products.
Our physicochemical analysis involves techniques such as liquid chromatography, mass spectrometry, spectroscopy, capillary electrophoresis, AUC, etc. These techniques enable the identification and quantification of critical components, impurities, and degradation in CGT products.

CASE STUDY

mRNA purity –AUC effectively distinguishes the different components in an mRNA sample, such as truncated mRNA (Peak 1), monomeric mRNA (Peak 2), long-chain mRNA, and mRNA aggregates (Peak 3& 4).

CASE STUDY
The Porton Advanced Characterization Platform utilizes LC-MS to analyze the capping ratio of the mRNA samples for the co-transcriptional capping reaction. Example:
The Rt 6.71 min in the left figure corresponds to the oligonucleotide fragments obtained by enzyme digestion. The 5′ capping ratio is obtained by quantifying the peak area of the capped or uncapped protein components.

Physicochemical and Characterization assays play a crucial role in the development and manufacturing of CGT. These assays encompass a wide range of techniques and testing that assess the physical and chemical attributes of therapeutic products. This includes determining the identity, purity, potency, stability, and structural integrity of CGT products.
Our physicochemical analysis involves techniques such as liquid chromatography, mass spectrometry, spectroscopy, capillary electrophoresis, AUC, etc. These techniques enable the identification and quantification of critical components, impurities, and degradation in CGT products.

CASE STUDY

mRNA purity –AUC effectively distinguishes the different components in an mRNA sample, such as truncated mRNA (Peak 1), monomeric mRNA (Peak 2), long-chain mRNA, and mRNA aggregates (Peak 3& 4).

CASE STUDY
The Porton Advanced Characterization Platform utilizes LC-MS to analyze the capping ratio of the mRNA samples for the co-transcriptional capping reaction. Example:
The Rt 6.71 min in the left figure corresponds to the oligonucleotide fragments obtained by enzyme digestion. The 5′ capping ratio is obtained by quantifying the peak area of the capped or uncapped protein components.

Physicochemical and Characterization assays play a crucial role in the development and manufacturing of CGT. These assays encompass a wide range of techniques and testing that assess the physical and chemical attributes of therapeutic products. This includes determining the identity, purity, potency, stability, and structural integrity of CGT products.
Our physicochemical analysis involves techniques such as liquid chromatography, mass spectrometry, spectroscopy, capillary electrophoresis, AUC, etc. These techniques enable the identification and quantification of critical components, impurities, and degradation in CGT products.

CASE STUDY

mRNA purity –AUC effectively distinguishes the different components in an mRNA sample, such as truncated mRNA (Peak 1), monomeric mRNA (Peak 2), long-chain mRNA, and mRNA aggregates (Peak 3& 4).

CASE STUDY
The Porton Advanced Characterization Platform utilizes LC-MS to analyze the capping ratio of the mRNA samples for the co-transcriptional capping reaction. Example:
The Rt 6.71 min in the left figure corresponds to the oligonucleotide fragments obtained by enzyme digestion. The 5′ capping ratio is obtained by quantifying the peak area of the capped or uncapped protein components.

Physicochemical and Characterization assays play a crucial role in the development and manufacturing of CGT. These assays encompass a wide range of techniques and testing that assess the physical and chemical attributes of therapeutic products. This includes determining the identity, purity, potency, stability, and structural integrity of CGT products.
Our physicochemical analysis involves techniques such as liquid chromatography, mass spectrometry, spectroscopy, capillary electrophoresis, AUC, etc. These techniques enable the identification and quantification of critical components, impurities, and degradation in CGT products.

CASE STUDY

mRNA purity –AUC effectively distinguishes the different components in an mRNA sample, such as truncated mRNA (Peak 1), monomeric mRNA (Peak 2), long-chain mRNA, and mRNA aggregates (Peak 3& 4).

CASE STUDY
The Porton Advanced Characterization Platform utilizes LC-MS to analyze the capping ratio of the mRNA samples for the co-transcriptional capping reaction. Example:
The Rt 6.71 min in the left figure corresponds to the oligonucleotide fragments obtained by enzyme digestion. The 5′ capping ratio is obtained by quantifying the peak area of the capped or uncapped protein components.

Physicochemical and Characterization assays play a crucial role in the development and manufacturing of CGT. These assays encompass a wide range of techniques and testing that assess the physical and chemical attributes of therapeutic products. This includes determining the identity, purity, potency, stability, and structural integrity of CGT products.
Our physicochemical analysis involves techniques such as liquid chromatography, mass spectrometry, spectroscopy, capillary electrophoresis, AUC, etc. These techniques enable the identification and quantification of critical components, impurities, and degradation in CGT products.

CASE STUDY

mRNA purity –AUC effectively distinguishes the different components in an mRNA sample, such as truncated mRNA (Peak 1), monomeric mRNA (Peak 2), long-chain mRNA, and mRNA aggregates (Peak 3& 4).

CASE STUDY
The Porton Advanced Characterization Platform utilizes LC-MS to analyze the capping ratio of the mRNA samples for the co-transcriptional capping reaction. Example:
The Rt 6.71 min in the left figure corresponds to the oligonucleotide fragments obtained by enzyme digestion. The 5′ capping ratio is obtained by quantifying the peak area of the capped or uncapped protein components.

Physicochemical and Characterization assays play a crucial role in the development and manufacturing of CGT. These assays encompass a wide range of techniques and testing that assess the physical and chemical attributes of therapeutic products. This includes determining the identity, purity, potency, stability, and structural integrity of CGT products.
Our physicochemical analysis involves techniques such as liquid chromatography, mass spectrometry, spectroscopy, capillary electrophoresis, AUC, etc. These techniques enable the identification and quantification of critical components, impurities, and degradation in CGT products.

CASE STUDY

mRNA purity –AUC effectively distinguishes the different components in an mRNA sample, such as truncated mRNA (Peak 1), monomeric mRNA (Peak 2), long-chain mRNA, and mRNA aggregates (Peak 3& 4).

CASE STUDY
The Porton Advanced Characterization Platform utilizes LC-MS to analyze the capping ratio of the mRNA samples for the co-transcriptional capping reaction. Example:
The Rt 6.71 min in the left figure corresponds to the oligonucleotide fragments obtained by enzyme digestion. The 5′ capping ratio is obtained by quantifying the peak area of the capped or uncapped protein components.

Physicochemical and Characterization assays play a crucial role in the development and manufacturing of CGT. These assays encompass a wide range of techniques and testing that assess the physical and chemical attributes of therapeutic products. This includes determining the identity, purity, potency, stability, and structural integrity of CGT products.
Our physicochemical analysis involves techniques such as liquid chromatography, mass spectrometry, spectroscopy, capillary electrophoresis, AUC, etc. These techniques enable the identification and quantification of critical components, impurities, and degradation in CGT products.

CASE STUDY

mRNA purity –AUC effectively distinguishes the different components in an mRNA sample, such as truncated mRNA (Peak 1), monomeric mRNA (Peak 2), long-chain mRNA, and mRNA aggregates (Peak 3& 4).

CASE STUDY
The Porton Advanced Characterization Platform utilizes LC-MS to analyze the capping ratio of the mRNA samples for the co-transcriptional capping reaction. Example:
The Rt 6.71 min in the left figure corresponds to the oligonucleotide fragments obtained by enzyme digestion. The 5′ capping ratio is obtained by quantifying the peak area of the capped or uncapped protein components.

Physicochemical and Characterization assays play a crucial role in the development and manufacturing of CGT. These assays encompass a wide range of techniques and testing that assess the physical and chemical attributes of therapeutic products. This includes determining the identity, purity, potency, stability, and structural integrity of CGT products.
Our physicochemical analysis involves techniques such as liquid chromatography, mass spectrometry, spectroscopy, capillary electrophoresis, AUC, etc. These techniques enable the identification and quantification of critical components, impurities, and degradation in CGT products.

CASE STUDY

mRNA purity –AUC effectively distinguishes the different components in an mRNA sample, such as truncated mRNA (Peak 1), monomeric mRNA (Peak 2), long-chain mRNA, and mRNA aggregates (Peak 3& 4).

CASE STUDY
The Porton Advanced Characterization Platform utilizes LC-MS to analyze the capping ratio of the mRNA samples for the co-transcriptional capping reaction. Example:
The Rt 6.71 min in the left figure corresponds to the oligonucleotide fragments obtained by enzyme digestion. The 5′ capping ratio is obtained by quantifying the peak area of the capped or uncapped protein components.

Physicochemical and Characterization assays play a crucial role in the development and manufacturing of CGT. These assays encompass a wide range of techniques and testing that assess the physical and chemical attributes of therapeutic products. This includes determining the identity, purity, potency, stability, and structural integrity of CGT products.
Our physicochemical analysis involves techniques such as liquid chromatography, mass spectrometry, spectroscopy, capillary electrophoresis, AUC, etc. These techniques enable the identification and quantification of critical components, impurities, and degradation in CGT products.

CASE STUDY

mRNA purity –AUC effectively distinguishes the different components in an mRNA sample, such as truncated mRNA (Peak 1), monomeric mRNA (Peak 2), long-chain mRNA, and mRNA aggregates (Peak 3& 4).

CASE STUDY
The Porton Advanced Characterization Platform utilizes LC-MS to analyze the capping ratio of the mRNA samples for the co-transcriptional capping reaction. Example:
The Rt 6.71 min in the left figure corresponds to the oligonucleotide fragments obtained by enzyme digestion. The 5′ capping ratio is obtained by quantifying the peak area of the capped or uncapped protein components.

Physicochemical and Characterization assays play a crucial role in the development and manufacturing of CGT. These assays encompass a wide range of techniques and testing that assess the physical and chemical attributes of therapeutic products. This includes determining the identity, purity, potency, stability, and structural integrity of CGT products.
Our physicochemical analysis involves techniques such as liquid chromatography, mass spectrometry, spectroscopy, capillary electrophoresis, AUC, etc. These techniques enable the identification and quantification of critical components, impurities, and degradation in CGT products.

CASE STUDY

mRNA purity –AUC effectively distinguishes the different components in an mRNA sample, such as truncated mRNA (Peak 1), monomeric mRNA (Peak 2), long-chain mRNA, and mRNA aggregates (Peak 3& 4).

CASE STUDY
The Porton Advanced Characterization Platform utilizes LC-MS to analyze the capping ratio of the mRNA samples for the co-transcriptional capping reaction. Example:
The Rt 6.71 min in the left figure corresponds to the oligonucleotide fragments obtained by enzyme digestion. The 5′ capping ratio is obtained by quantifying the peak area of the capped or uncapped protein components.

Physicochemical and Characterization assays play a crucial role in the development and manufacturing of CGT. These assays encompass a wide range of techniques and testing that assess the physical and chemical attributes of therapeutic products. This includes determining the identity, purity, potency, stability, and structural integrity of CGT products.
Our physicochemical analysis involves techniques such as liquid chromatography, mass spectrometry, spectroscopy, capillary electrophoresis, AUC, etc. These techniques enable the identification and quantification of critical components, impurities, and degradation in CGT products.

CASE STUDY

mRNA purity –AUC effectively distinguishes the different components in an mRNA sample, such as truncated mRNA (Peak 1), monomeric mRNA (Peak 2), long-chain mRNA, and mRNA aggregates (Peak 3& 4).

CASE STUDY
The Porton Advanced Characterization Platform utilizes LC-MS to analyze the capping ratio of the mRNA samples for the co-transcriptional capping reaction. Example:
The Rt 6.71 min in the left figure corresponds to the oligonucleotide fragments obtained by enzyme digestion. The 5′ capping ratio is obtained by quantifying the peak area of the capped or uncapped protein components.

Physicochemical and Characterization assays play a crucial role in the development and manufacturing of CGT. These assays encompass a wide range of techniques and testing that assess the physical and chemical attributes of therapeutic products. This includes determining the identity, purity, potency, stability, and structural integrity of CGT products.
Our physicochemical analysis involves techniques such as liquid chromatography, mass spectrometry, spectroscopy, capillary electrophoresis, AUC, etc. These techniques enable the identification and quantification of critical components, impurities, and degradation in CGT products.

CASE STUDY

mRNA purity –AUC effectively distinguishes the different components in an mRNA sample, such as truncated mRNA (Peak 1), monomeric mRNA (Peak 2), long-chain mRNA, and mRNA aggregates (Peak 3& 4).

CASE STUDY
The Porton Advanced Characterization Platform utilizes LC-MS to analyze the capping ratio of the mRNA samples for the co-transcriptional capping reaction. Example:
The Rt 6.71 min in the left figure corresponds to the oligonucleotide fragments obtained by enzyme digestion. The 5′ capping ratio is obtained by quantifying the peak area of the capped or uncapped protein components.

Physicochemical and Characterization assays play a crucial role in the development and manufacturing of CGT. These assays encompass a wide range of techniques and testing that assess the physical and chemical attributes of therapeutic products. This includes determining the identity, purity, potency, stability, and structural integrity of CGT products.
Our physicochemical analysis involves techniques such as liquid chromatography, mass spectrometry, spectroscopy, capillary electrophoresis, AUC, etc. These techniques enable the identification and quantification of critical components, impurities, and degradation in CGT products.

CASE STUDY

mRNA purity –AUC effectively distinguishes the different components in an mRNA sample, such as truncated mRNA (Peak 1), monomeric mRNA (Peak 2), long-chain mRNA, and mRNA aggregates (Peak 3& 4).

CASE STUDY
The Porton Advanced Characterization Platform utilizes LC-MS to analyze the capping ratio of the mRNA samples for the co-transcriptional capping reaction. Example:
The Rt 6.71 min in the left figure corresponds to the oligonucleotide fragments obtained by enzyme digestion. The 5′ capping ratio is obtained by quantifying the peak area of the capped or uncapped protein components.

Physicochemical and Characterization assays play a crucial role in the development and manufacturing of CGT. These assays encompass a wide range of techniques and testing that assess the physical and chemical attributes of therapeutic products. This includes determining the identity, purity, potency, stability, and structural integrity of CGT products.
Our physicochemical analysis involves techniques such as liquid chromatography, mass spectrometry, spectroscopy, capillary electrophoresis, AUC, etc. These techniques enable the identification and quantification of critical components, impurities, and degradation in CGT products.

CASE STUDY

mRNA purity –AUC effectively distinguishes the different components in an mRNA sample, such as truncated mRNA (Peak 1), monomeric mRNA (Peak 2), long-chain mRNA, and mRNA aggregates (Peak 3& 4).

CASE STUDY
The Porton Advanced Characterization Platform utilizes LC-MS to analyze the capping ratio of the mRNA samples for the co-transcriptional capping reaction. Example:
The Rt 6.71 min in the left figure corresponds to the oligonucleotide fragments obtained by enzyme digestion. The 5′ capping ratio is obtained by quantifying the peak area of the capped or uncapped protein components.

Physicochemical and Characterization assays play a crucial role in the development and manufacturing of CGT. These assays encompass a wide range of techniques and testing that assess the physical and chemical attributes of therapeutic products. This includes determining the identity, purity, potency, stability, and structural integrity of CGT products.
Our physicochemical analysis involves techniques such as liquid chromatography, mass spectrometry, spectroscopy, capillary electrophoresis, AUC, etc. These techniques enable the identification and quantification of critical components, impurities, and degradation in CGT products.

CASE STUDY

mRNA purity –AUC effectively distinguishes the different components in an mRNA sample, such as truncated mRNA (Peak 1), monomeric mRNA (Peak 2), long-chain mRNA, and mRNA aggregates (Peak 3& 4).

CASE STUDY
The Porton Advanced Characterization Platform utilizes LC-MS to analyze the capping ratio of the mRNA samples for the co-transcriptional capping reaction. Example:
The Rt 6.71 min in the left figure corresponds to the oligonucleotide fragments obtained by enzyme digestion. The 5′ capping ratio is obtained by quantifying the peak area of the capped or uncapped protein components.

Physicochemical and Characterization assays play a crucial role in the development and manufacturing of CGT. These assays encompass a wide range of techniques and testing that assess the physical and chemical attributes of therapeutic products. This includes determining the identity, purity, potency, stability, and structural integrity of CGT products.
Our physicochemical analysis involves techniques such as liquid chromatography, mass spectrometry, spectroscopy, capillary electrophoresis, AUC, etc. These techniques enable the identification and quantification of critical components, impurities, and degradation in CGT products.

CASE STUDY

mRNA purity –AUC effectively distinguishes the different components in an mRNA sample, such as truncated mRNA (Peak 1), monomeric mRNA (Peak 2), long-chain mRNA, and mRNA aggregates (Peak 3& 4).

CASE STUDY
The Porton Advanced Characterization Platform utilizes LC-MS to analyze the capping ratio of the mRNA samples for the co-transcriptional capping reaction. Example:
The Rt 6.71 min in the left figure corresponds to the oligonucleotide fragments obtained by enzyme digestion. The 5′ capping ratio is obtained by quantifying the peak area of the capped or uncapped protein components.

Physicochemical and Characterization assays play a crucial role in the development and manufacturing of CGT. These assays encompass a wide range of techniques and testing that assess the physical and chemical attributes of therapeutic products. This includes determining the identity, purity, potency, stability, and structural integrity of CGT products.
Our physicochemical analysis involves techniques such as liquid chromatography, mass spectrometry, spectroscopy, capillary electrophoresis, AUC, etc. These techniques enable the identification and quantification of critical components, impurities, and degradation in CGT products.

CASE STUDY

mRNA purity –AUC effectively distinguishes the different components in an mRNA sample, such as truncated mRNA (Peak 1), monomeric mRNA (Peak 2), long-chain mRNA, and mRNA aggregates (Peak 3& 4).

CASE STUDY
The Porton Advanced Characterization Platform utilizes LC-MS to analyze the capping ratio of the mRNA samples for the co-transcriptional capping reaction. Example:
The Rt 6.71 min in the left figure corresponds to the oligonucleotide fragments obtained by enzyme digestion. The 5′ capping ratio is obtained by quantifying the peak area of the capped or uncapped protein components.

Physicochemical and Characterization assays play a crucial role in the development and manufacturing of CGT. These assays encompass a wide range of techniques and testing that assess the physical and chemical attributes of therapeutic products. This includes determining the identity, purity, potency, stability, and structural integrity of CGT products.
Our physicochemical analysis involves techniques such as liquid chromatography, mass spectrometry, spectroscopy, capillary electrophoresis, AUC, etc. These techniques enable the identification and quantification of critical components, impurities, and degradation in CGT products.

CASE STUDY

mRNA purity –AUC effectively distinguishes the different components in an mRNA sample, such as truncated mRNA (Peak 1), monomeric mRNA (Peak 2), long-chain mRNA, and mRNA aggregates (Peak 3& 4).

CASE STUDY
The Porton Advanced Characterization Platform utilizes LC-MS to analyze the capping ratio of the mRNA samples for the co-transcriptional capping reaction. Example:
The Rt 6.71 min in the left figure corresponds to the oligonucleotide fragments obtained by enzyme digestion. The 5′ capping ratio is obtained by quantifying the peak area of the capped or uncapped protein components.

Physicochemical and Characterization assays play a crucial role in the development and manufacturing of CGT. These assays encompass a wide range of techniques and testing that assess the physical and chemical attributes of therapeutic products. This includes determining the identity, purity, potency, stability, and structural integrity of CGT products.
Our physicochemical analysis involves techniques such as liquid chromatography, mass spectrometry, spectroscopy, capillary electrophoresis, AUC, etc. These techniques enable the identification and quantification of critical components, impurities, and degradation in CGT products.

CASE STUDY

mRNA purity –AUC effectively distinguishes the different components in an mRNA sample, such as truncated mRNA (Peak 1), monomeric mRNA (Peak 2), long-chain mRNA, and mRNA aggregates (Peak 3& 4).

CASE STUDY
The Porton Advanced Characterization Platform utilizes LC-MS to analyze the capping ratio of the mRNA samples for the co-transcriptional capping reaction. Example:
The Rt 6.71 min in the left figure corresponds to the oligonucleotide fragments obtained by enzyme digestion. The 5′ capping ratio is obtained by quantifying the peak area of the capped or uncapped protein components.














